
“How Do You Really Know?” – 19 Questions Every Healthcare IT Leader Should Ask About…



TRUSTED BY VENDORS, LABORATORIES AND HEALTH SYSTEMS WORLDWIDE
Meet Cymetryc
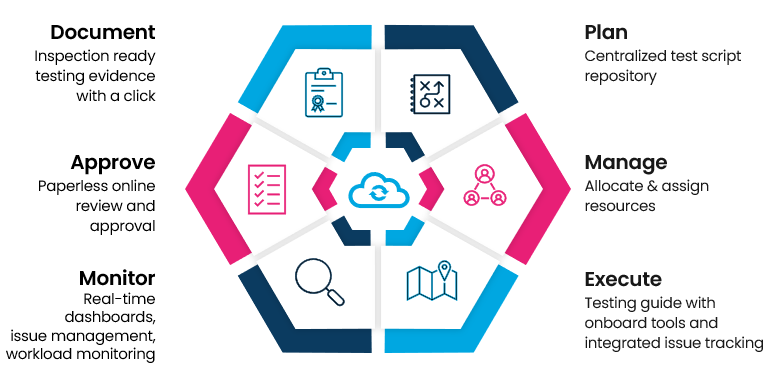
Whatever your testing need, Cymetryc has you covered! Conquer user acceptance testing (UAT), validation testing, functional testing, end-to-end testing, Epic quarterly upgrade testing, Beaker implementations (including CCV), instrument testing, analyzer wet testing, or any upstream/downstream application testing
RESOURCES & TEST MANAGEMENT
Ditch the shared spreadsheet!
Build, plan, and assign testing. Review workloads, manage testing hand-offs. Dashboards deliver real-time testing and review status.
EASY EXECUTION & EVIDENCE CAPTURE
No more snipping tools!
Empower testers with guided test execution, integrated screen captures and easy evidence collection. One-click issue reporting ensures defects are reported and fully documented. And best of all, Cymetryc is completely application agnostic so you can finally standardize your all your application testing.
PAPERLESS REVIEW AND APPROVAL
No more binders!
Staff can easily examine and digitally approve testing results as needed in the web-based portal. Reviews can be done anytime and anywhere, with complete visibility to all captured images, test case details and tester notes.
TESTING EVIDENCE PERFECTED
No more wasted time formatting proof of testing!
Prove quality and compliance with easy to generate inspection-ready reports that are consistent regardless of application or team. Whether testing detail, testing summary, or requirements mapping, Cymetryc's self documenting system provides picture-perfect testing evidence on demand.
Powerful Purpose-Built Platform
Discover the most consistent, comprehensive, and cost-effective way to test all your third party applications.
Ready to ditch the spreadsheet?
Application Agnostic
Regardless of application, workflow, or team, provides a single testing methodology
Paperless Review & Approvals
No more mountains of paper. Review & approve from anywhere you have browser access.
Built-In Compliance Best Practices
So you always exceed quality &
regulatory requirements



Meet Cymetryc
Increased Visibility
See the full testing and review scope real time. Measure progress, view workloads and manage issues.
Manage Resource
Allocations
See your testing status and identify who and where are the hold-ups.
Save Time & Effort
Focus your users on actual testing and learning new features. Don’t waste their time fighting with documents, spread sheets, or status updates. Let Cymetryc dynamically conquer that for them.
BLOG & Insights